This work expands the knowledge on zinc electrode kinetics over a wide range of pH and augments the existing literature on zinc corrosion. In alkaline conditions corrosion occurs chiefly by the formation of zinc hydroxide complexes or zinc oxides that could protect the surface depending on local pH and potential at the metal surface. The surface oxides thermodynamically predicted to form in this pH (7 to 10) do not form an effective corrosion protection barrier. In the pH range (7 to 10), the lowered cathodic reaction rate reduces overall zinc corrosion rates. In acidic solutions, the active form of corrosion occurring is controlled by the kinetics of the cathodic reaction (predominantly hydrogen evolution). The analysis shows that, in general, variations in corrosion of zinc with pH are associated with the cathodic currents. The electrochemical data was then contrast to thermodynamic chemical equilibrium diagrams. Zinc is the 24th-most abundant element in the Earth's crust.Anodic and cathodic polarization of zinc was carried out in 0.1 M sodium chloride (NaCl) solution with pH ranging from 1 to 13.Zinc is easily recycled and about 30% of the zinc produced annually is recycled metal. Almost all mined zinc (95%) comes from zinc sulfide ore.Foremost among these is brass, an alloy of copper and zinc. The product is a layer of zinc carbonate, which inhibits further degradation, thus protecting the metal beneath it. Although galvanization is used to protect metals against corrosion, zinc actually does tarnish in air.Zinc, its oxide, and other compounds are found in batteries, sunscreen, paints, and other products. Brass and bronze production account for another 17% of zinc's usage. Of the 12 million tons of the metal produced annually, about half goes to galvanization. 
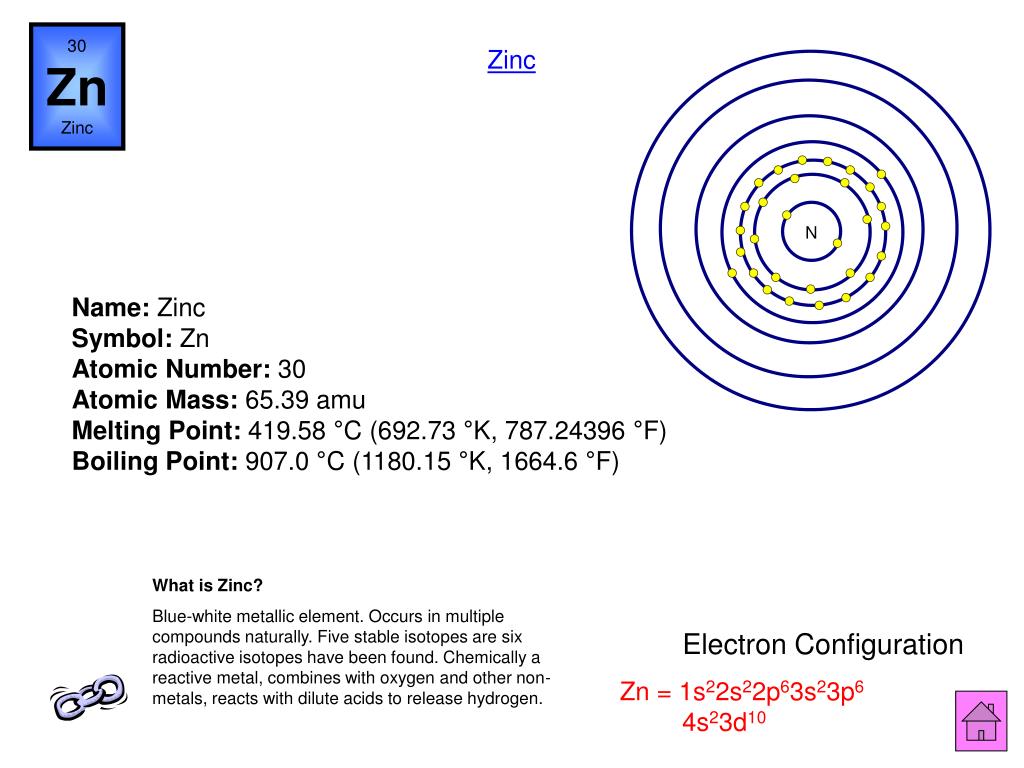
It is the fourth-most common metal for industry, after iron, aluminum, and copper. Problems from excessive ingestion of zinc lozenges or from industrial exposure to zinc have also been reported. The FDA has issued warnings regarding zinc nasal sprays and swabs. One noteworthy side effect of excessive zinc exposure is a permanent loss of smell and/or taste. Ingesting coins containing zinc has been known to cause death, as the metal reacts with gastric juice, corroding the gastrointestinal tract and producing zinc intoxication. While it's important to get enough zinc, too much can cause problems-including suppressing the absorption of iron and copper.Foods rich in zinc include lean meat and seafood. Zinc deficiency may also be a causal factor in age-related vision deterioration. The mineral is important for immune function, white blood cell formation, egg fertilization, cell division, and a host of other enzymatic reactions. It is the second-most abundant metal in the body, after iron. Zinc is an essential mineral for human health.Because zinc is often found with copper, the metal's use was more common as an alloy rather than as a pure element. However, artifacts do exist proving its early use, including a sheet of Athenian zinc, dating back to 300 BCE. Although zinc was used by the ancient Greeks and Romans, it was not as common as iron or copper, probably because the element boils away prior to reaching the temperature required to extract it from ore.According to the International Zinc Association (ITA), zinc was recognized as a unique substance in India by 1374 and is believed to have been discovered by Indian metallurgists before 1000 BCE. The micro algal cell wall has indeed the capacity to bind with these ions via its negatively charged moieties 5. While Champion may have been the first to isolate zinc, smelting of the element had been in practice in India since the 9th century BCE. mention that the pH and the cell’s wall play by themselves one of the most important functions in the adsorption of heavy metals by micro-algal cells. However, English metallurgist William Champion had actually patented his process for isolating zinc several years earlier. Andreas Marggraf is credited with isolating the element zinc in 1746, by heating calamine ore and carbon together in a closed vessel. The element name is believed to come from the German word "zinke," which means "pointed." This is likely a reference to the pointed zinc crystals that form after zinc is smelted. Paracelsus, a Swiss-born, German Renaissance physician, alchemist, and astrologer, is credited with giving zinc its name.Sometimes zinc is considered to be a post-transition metal. Zinc has the element symbol Zn and atomic number 30, making it a transition metal and the first element in Group 12 of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
